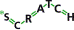
Soon an excipient was added, which enhanced efficacy and shortened time to onset of efficacy.
In April 2008 a complete new registration followed as a highly effective drug combination, indicated for all areas of shortages and deficiencies in Pharmacovigilance. Optimisations by new ingredients and excipients as well as by new indications in the field of medical devices have occurred by 2015. Efficacy, tolerance, and general applicability have been sustainably enhanced. A positive benefit-risk ratio has been demonstrated over and over again.
Safety Competence
Risk Assessment
Training and
Consulting with Heart.

